Franchise at IILS
Bridging the Gap Between Academia and Industry in Clinical Research
Establish a premium, industry-integrated learning centre in your city with
Iqva Institute of Life Science.

Iqva Institute of Life Science offers a unique opportunity to establish a professionally driven clinical research learning centre across India, built on strong academic foundations and real industry integration. With a clear roadmap to establish presence in 21 cities, the initiative is built to deliver structured, industry-aligned learning through a centrally managed system powered by IQVAMED.

- 21, Cities | PAN India Presence
- 21 Exclusive City Partnerships

- Central Academic Control
- LMS-Based Delivery

- 21, Cities | PAN India Presence
- 21 Exclusive City Partnerships

- Clinical Ecosystem Focus
- Outcome-Oriented Training
Why IILS Learning Center Across India?
Across the country, clinical trials are conducted at a wide network of government and private hospitals, as required by regulatory frameworks to ensure regional diversity and representation. However, one of the most critical challenges faced by sponsors and CROs is the availability of skilled clinical research professionals at the site level.
This challenge becomes even more significant in a diverse country like India, where multiple regional languages and local patient interactions play a vital role in study conduct. Ethical requirements such as informed consent demand clear communication in the patient’s native language, making it essential to have trained professionals who understand both clinical protocols and regional contexts.
With a focused vision to establish professionally driven clinical research learning centres across 21 key cities, Iqva Institute of Life Science addresses this gap by developing industry-ready talent aligned with local needs. The model is designed not just to educate, but to create a skilled workforce capable of supporting clinical research activities effectively across diverse regions of India.
Role of the Centre Partner

Center Setup & Infrastructure

Student Acquisition & Admissions

Classroom Management

Sessions Management

Faculty Acquisition | Local Hiring

70%: Local faculty (offline)

30%: Central expert sessions (live/recorded)

Opportunity for Life Science Students
Graduates from life sciences and healthcare backgrounds possess a strong foundation to enter the clinical research domain. Individuals holding degrees in Life Sciences, Pharmacy, Medicine, Dentistry, Nursing, Biotechnology, Microbiology, Biochemistry, and allied disciplines are well-positioned to explore diverse and evolving roles within this field.
Clinical research extends beyond traditional boundaries, offering opportunities across multiple specialized functions such as:

Drug Development

Regulatory Affairs

Quality Assurance

Clinical Data Management

eTMF & Documentation
A wide spectrum of career opportunities across the clinical research and healthcare ecosystem.
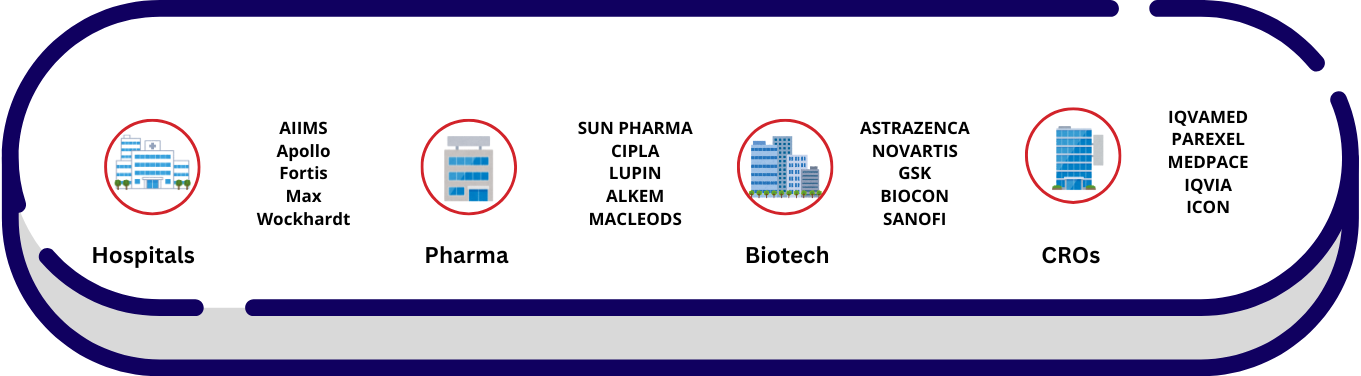
Workspaces of CR Personnel
Graduates trained and certified through Iqva Institute of Life Science are prepared to explore opportunities across a wide network of clinical research organizations, pharmaceutical companies, and hospital-based research sites.
With the increasing demand for skilled professionals at the site level, particularly in diverse regions across India, certified candidates are well-positioned to contribute to roles that require both technical knowledge and regional adaptability.
Academic Programs Offered by IILS Centres:
1 Year
PG Diploma in Clinical Research (PGDCR)
6 Month
Professional Diploma in Clinical Research (PDCR)
1 Year
PG Diploma in Pharmacovigilance (PGDPV)
6 Month
Professional Diploma in Pharmacovigilance (PDPV)
1 Year
PG Diploma in Regulatory Affairs (PGDRA)
Your Centre, Operational in 5 Phases
Phase 1:
Initiation

Phase 2:
Planning & Setup

Phase 3:
Training & Preparation

Phase 2:
Pre-Launch

Phase 5:
Launch

From Agreement to Launch – A 45-Days Centre Establishment | Operational

Day 01 – Agreement & Onboarding
Formalization of partnership and initiation of the onboarding process.

Day 05 – Infrastructure Planning
Finalization of centre layout, branding elements, and interior execution plans.
Day 10 – Recruitment Activation
Release of hiring requirements for faculty and support staff.

Day 10–20 – Recruitment & Finalization
Screening, interviews, and selection of candidates in coordination with IILS.

Day 21–25 – Faculty Induction & Training
Structured onboarding and training programs conducted by Iqva Institute of Life Science.
Day 30 – Academic & Marketing Assets
Access to course materials, LMS, and promotional resources.

Day 35 – Pre-Launch Activities Begin
Local outreach, branding, and student engagementactivities initiated.

Day 40 – Launch Campaign Activation
Strategic marketing rollout and awareness campaigns.

Day 45 – Official Centre Launch
Formal inauguration, press coverage, and commencement of operations.
Investment Overview
Iqva Institute of Life Science invites strategic partners who bring financial capability, long-term vision, and a commitment to delivering high-quality education in the clinical research and healthcare domains.
The model is designed for individuals or organizations prepared to establish and manage a professionally driven learning centre, aligned with the centralized academic and operational framework of IILS.
-
Operational Commitment
- Ability to mobilize funds through internal or external sources to support centre establishment and operations. -
Operational Commitment
- Willingness to independently manage at least 70% of operational expenses, ensuring stability and continuity. -
City-Based Opportunity
- Franchise and Master Franchise partnerships are being offered across 21 strategically selected cities in India. -
Flexible Investment Structure
- Total investment may vary based on location, city tier, centre size, and overall operational scope.
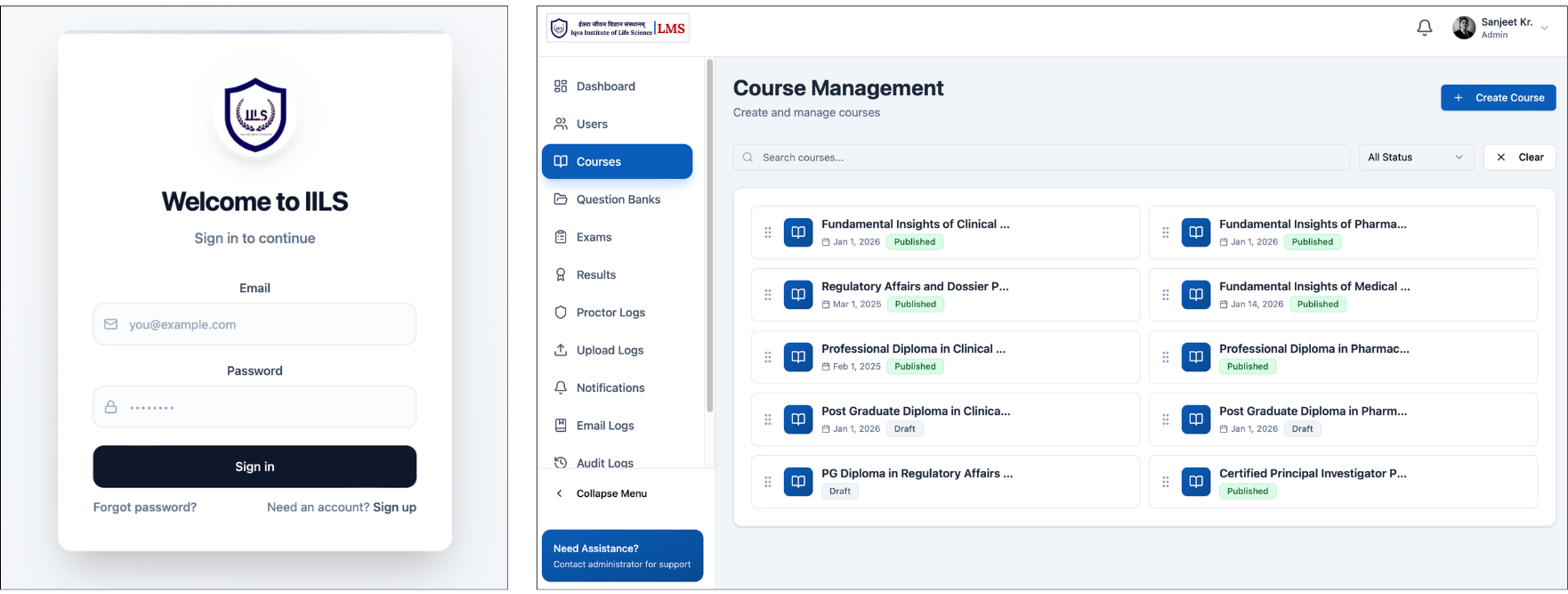
IILS Learning Management System (LMS)
Centrally Managed Learning Ecosystem : All academic programs and training sessions are centrally designed, managed, and delivered through the Learning Management System of Iqva Institute of Life Science.
The LMS ensures consistency in curriculum delivery, structured session planning, and seamless coordination across all centres, regardless of location. It enables real-time access to course materials, session schedules, and academic resources, maintaining uniform quality standards throughout the network.
Faculty and Staff Framework
The academic delivery across all centres of Iqva Institute of Life Science is supported by a carefully selected and trained faculty network. While faculty may be sourced locally (70 %), all candidates must possess a minimum of five years of relevant industry experience in their respective domain and undergo a standardized evaluation, approval, and training process to ensure alignment with IILS academic standards.
This hybrid model combines local accessibility with central academic control, ensuring consistency in teaching quality, domain expertise, and professional delivery across all regions.

Faculty Selection & Screening by IILS

Industry-Experienced Professionals

Domain-Specific Expertise

Trained & Standardized Delivery

Consistent Performance & Evaluation

WHY CHOOSE AN IILS FRANCHISE?

Centrally Controlled Academic Framework
All programs, sessions, and certifications are managed through a unified LMS, ensuring consistent quality across every centre.

Built to Address Real Industry Gaps
Designed to bridge the shortage of skilled clinical research professionals, especially at site levels across diverse regions.
Regionally Scalable & Standardized Model
Structured to operate efficiently across multiple cities while maintaining uniform academic and operational standards.
Exclusive City-Based Expansion Strategy
Limited partnerships across 21 cities ensure focused growth, strong positioning, and reduced internal competition.
Expansion Roadmap Across 21 Strategic Cities in India
West India
- 1.Navi Mumbai
- 2.Pune
- 3.Nashik
- 4.Nagpur
- 5.Ahmedabad
North India
- 6.Chandigarh
- 7.Jaipur
- 8.Jodhpur
- 9.Noida
- 10.Gurugram
- 11.Lucknow
- 12.Rishikesh
- 13.Bathinda
Central India
- 14.Bhopal
East India
- 14.Bangalore
- 15.Patna
- 16.Kolkata
- 17.Bhubaneswar
- 18.Ranchi
South India
- 19.Hyderabad
- 20.Bangalore
Union Territory
- 21.Vijaypur
What IILS Provides to Centre Partners

Centrally Designed Academic Programs

Standardized Curriculum & Certification Framework

Learning Management System (LMS) Access

Structured Session Planning & Delivery System

Faculty Screening & Final Approval

Faculty Training & Induction Programs

Continuous Academic Monitoring & Evaluation

Quality Assurance & Academic Audits

Complete Study Materials & Content Library

Presentation Decks & Teaching Resources

Access to Updated Industry-Relevant Content

Centralized Content Upgradation & Control

Branding Guidelines & Identity Support

Marketing Strategy & Campaign Frameworks

Admissions Process & Counseling Support

Operational SOPs & Centre Management Guidance
Looking for a Franchise Opportunity?
📩 Collaborate With Us
Colleges, institutions, and strategic partners looking to integrate industry-aligned clinical research programs and establish dedicated learning centres may reach out to us. Explore Collaboration | info@iilsc.in